Ammonia (NH3) synthesis from nitrogen (N2) and hydrogen (H2) is one of the most important chemical reactions in modern industry. Because of the high stability of the N≡N triple bond in N2, industrial ammonia production mainly relies on the Haber-Bosch process, which converts N2 and H2 to NH3 under high temperatures (350 - 500 ℃) and high pressures (10 - 30 MPa). This process is highly energy-intensive and associated with substantial carbon emissions.
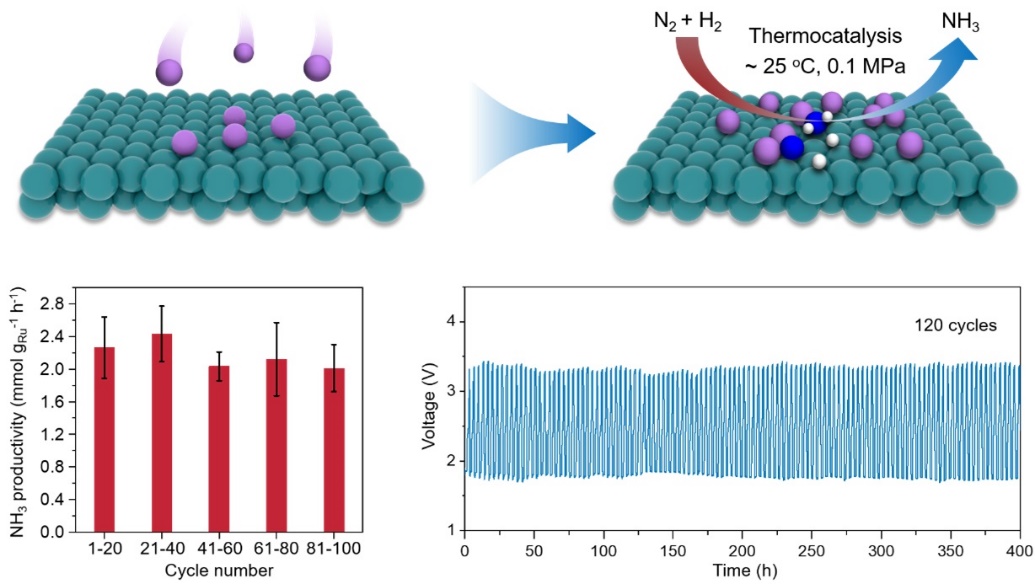
Schematic illustration and the performance of the thermocatalytic ambient-condition ammonia synthesis from N2 and H2 at the metallic Li/Ru interfaces (Image by HOU Ying)
In a study published in Chem, a research team led by Prof. DENG Dehui and Prof. YU Liang from the State Key Laboratory of Catatalysis(SKLC), in collaboration with Prof. CUI Yi from the Suzhou Institute of Nano-Tech and Nano-Bionics of CAS, developed a novel catalyst by depositing metallic lithium (Li) on ruthenium (Ru) surface to form highly active Li/Ru interfaces. This catalyst enables thermocatalytic conversion of N2 and H2 to NH3 at room temperature and ambient pressure.
The researchers found that the Li/Ru interface exhibited a synergetic effect that promoted both N2 activation and hydrogenation steps. Electron donation from Li to the antibonding orbital of adsorbed N2 facilitated N2 dissociation, while Li-N bonding interaction favored the hydrogenation of NHx intermediates.
The researchers further demonstrated this ambient-condition ammonia synthesis in a reversible Li battery (RLB), with Li metal as the anode and well-dispersed Ru nanoparticles on carbon nanotubes (Ru/CNTs) as the cathode. They found that Li/Ru interfaces are generated in situ during battery discharge. With a mixture of N2 and H2 flowing across the Li/Ru interface on the cathode, an NH3 productivity of 2.43 mmolNH3 gRu-1 h-1 was achieved at about 25 ℃ and 0.1 MPa. Moreover, through charge-discharge cycling of the RLB to enable the in situ generation and regeneration of the Li/Ru interfaces, this process operated stably for over 400 hours across more than 120 cycles.
"Integrated with high-efficiency energy-storage RLB systems, this process opens a new route for establishing a low-energy and sustainable paradigm for NH3 synthesis," said Prof. DENG.